Pharmaceuticals
Advance your Pharma 4.0 journey. Boost efficiency, increase productivity and improve access to medicine.
Watch Video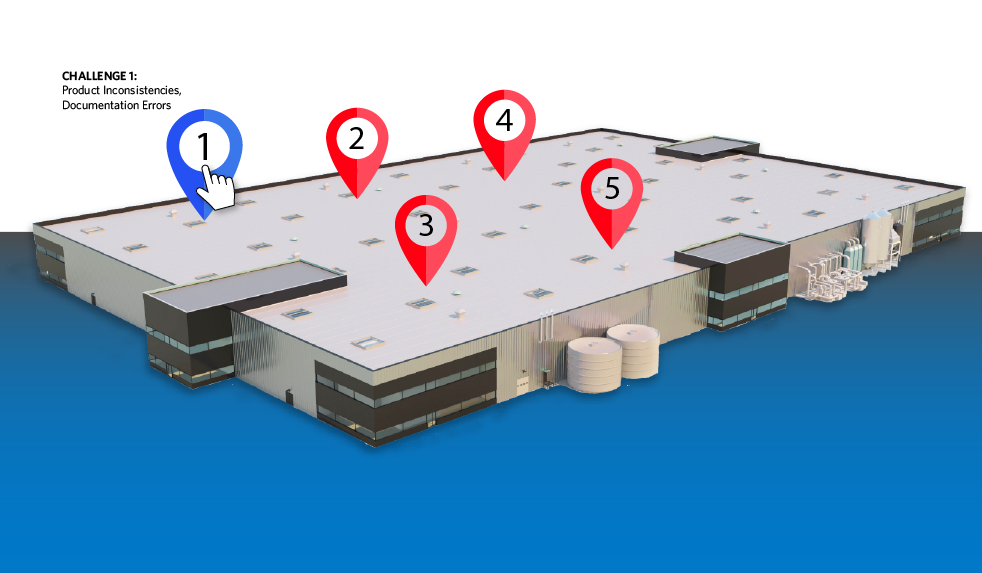
Navigate 5 Common Pharma Challenges
See how AspenTech can accelerate your digital transformation in pharma, boosting efficiency and productivity—and improving access to medicine.
-
■Gain time on the patent clock with shorter development times
■Empower Quality by Design (QbD) with rigorous process simulation for optimized processes
■Combine diverse data to discover new insights
■Realize improvements in product quality, yield and throughput
.png?h=415&w=675&la=ru&hash=2D5117BB0E3522DDD614E67EB64146AD)
Accelerate Tech Transfer and Ensure Quality Through a Digital Twin Strategy
The Digital Twin presents several advantages — learn more from Robert Zuban.
Blog
Ferring Prototypes Continuous Manufacturing with Advanced PAT and Closed-Loop Control, Potential for Dramatically Lower COGS
Ferring Pharmaceuticals, a multinational biopharmaceutical company, needed a solution that would enable faster commercialization, more cost-efficient production and reduced supply lead time while main...
Case Study
Pfizer Accelerates Process Design and Scale Up Using aspenONE® Engineering
Pfizer enhances new process development by coupling computational exercises with experiment planning, execution and revision. Learn how the visualization of process design can guide experimentation an...
On-Demand Webinar-
■Accelerate production execution and release with full cGMP compliance
■Digitalize paper-based manufacturing with procedural automation and electronic batch records
■Ensure data integrity and transparency per FDA 21 CFR Part 11 and EU Annex 11

Accelerate Your Pharma 4.0 Journey with Our Manufacturing Execution Systems (MES) Solution
Today’s pharmaceutical manufacturers face increasing pressure to deliver high-quality medicines quickly, efficiently and economically while complying with regulatory requirements and meeting sustainab...
Video
Digitalize Batch Execution and Records With Aspen Production Execution Manager™
How are you keeping up with growing demand? Digitalize batch execution and records with Aspen Production Execution Manager™ to accelerate time-to-market with accuracy while maintaining regulatory comp...
Interactive Infographic
Progress Your Pharma 4.0 Journey by Solving Industrial DataOps Challenges
As the volume of data within your operations continues to grow, it becomes increasingly difficult to integrate, manage and govern this data. For many companies, it remains one of the biggest challenge...
On-Demand Webinar-
■Achieve sustainability targets with 2021 Green Supply Chain Award winning models
■Address daily shifts in supply and demand with agility and confidence
■Balance near and long-term planning and scheduling priorities
■Build realistic schedules for improved on-target delivery

Riding the Roller Coaster of Pharma Production Scheduling
Managing the master schedule for pharmaceutical production is like riding a roller coaster — with both calm periods and high-velocity swings. It requires making complex decisions daily in response to ...
On-Demand Webinar
Navigating Pharma Scheduling Challenges During Periods of Volatility and Supply Chain Disruptions
In this interview, Cade VanRooyen describes the complex decisions master schedulers face and how digital scheduling solutions can significantly improve the consistent on-time delivery of life-saving m...
Interview with an Expert
How Industrial AI & Next-Generation Data Management Support Future-Proofing the Supply Chain
Building resilient and future-proofed supply chains require implementing technology that can bring people and processes together. Industrial AI can play a role in both alleviating current supply chain...
Article-
■Predict equipment degradation and impending failures early with a 2021 Breakthrough Product
■Minimize false alarms and failures with high prediction accuracy
■Deploy quickly and easily scale from site to enterprise
■Realize immediate impact within weeks, not months

GSK Creates a Future-Ready Supply Chain with Predictive and Prescriptive Maintenance
GlaxoSmithKline (GSK) implemented Aspen Mtell® to increase the robustness of their pharma supply chains by preventing unplanned production downtime.
Case Study
Making Maintenance a True Asset in Pharma Manufacturing Through Digitalization
Did you know that more than 80% of equipment failures in manufacturing are caused by human error or adverse operating conditions rather than time-based wear and tear? That’s why pharmaceutical compani...
On-Demand Webinar
Maximize Supply Chain Resiliency with Optimal Downtime Scheduling
Achieve resilient operations by evaluating optimized downtime scheduling options to minimize impact on production and order commitments. Watch this video to learn how to leverage prescriptive maintena...
Video